| 1. Count the total valence electrons in the molecule. Add electrons for negative charge. Subtract electrons for positive charge. |
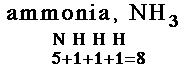 |
|
2. Place the least electronegative atom in the center. (Except hydrogen.)
Go to Electronegativities chart. |
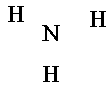 |
| 3. Connect the outer atoms to the central one with single bonds. Add electron pairs as needed to form octets. (Except hydrogen.) |
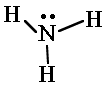 |
| 4. Count the electrons you have. If there are too many, replace two electron pairs with a single bond until you have the correct number of valence electons. |
3 bonds x 2 = 6 1 lone pair x 2 = 2 total is 8. Valence is 8. Done. |
| 5. Check that each atom (except hydrogen) has four electron pairs. | H two electrons. N eight electrons. Check. |