If you stress a system at equilibrium, the equilibrium will change to relieve the stress.
PCl5(g) 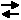 PCl3(g) + Cl2(g) + 92.5 kJ
Left side ------------------------Right side PCl3(g) + Cl2(g) + 92.5 kJ
Left side ------------------------Right side
- Concentration
Raise the concentration of Cl2 by adding more of it to the system. Which way does the equilibrium shift to reduce the Cl2 we added?
- Volume
Apply pressure and squeeze the system down to a smaller volume. Which side would have fewer moles of gas?
- Temperature
We raise the temperature by adding energy to the system. Which way will the system shift to use up some of this energy?
|