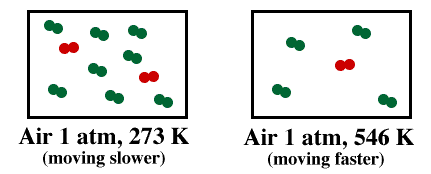
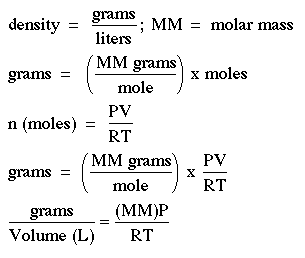
Now consider a hot air balloon at sea level with the pressure at 1 atm. Assume a hot air balloon displaces 500 cubic meters of air. A cubic meter is 1000 L. How many moles of air are displaced if the surrounding air is 20oC? How many moles of air are inside the balloon if the air is heated to 80oC? If the average molar mass of air is 28.8 g/mol, compute the density of air inside and outside of the balloon. Density = g/L If the "lift" of the balloon is equal to the mass of the air displaced, how much can the balloon lift? (For net lift you have to subtract the mass of the balloon and the air it holds.) |