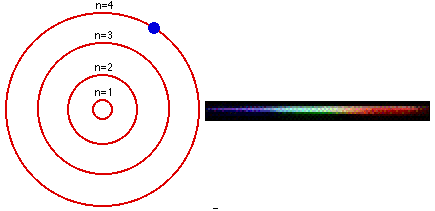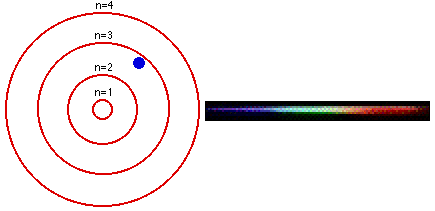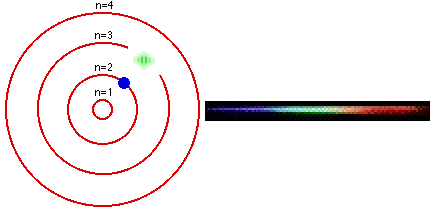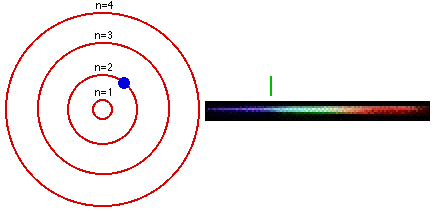
 1. The electron in a hydrogen atom can have only certain fixed energy values. 2. Electrons cannot drop below their lowest, or GROUND state. For hydrogen this is n=1. 3. If an electron is given energy it is EXCITED and can move up to a higher level. 4. When an electron drops between levels, a photon is given off with the energy difference. |